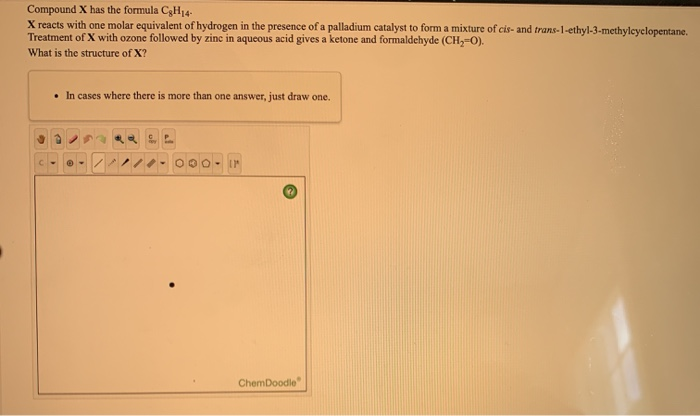
25 moles which gives us 400 g per mole of the compound. We simply need to take the grams and the moles that were provided in this problem and divide them. To calculate the molar mass which has units of grams of the compound, per moles of the compound. And they give us some information associated with the moles of each compound and the corresponding masses. Now, for part two we have two imaginary compounds, X cl two and Y C L two. 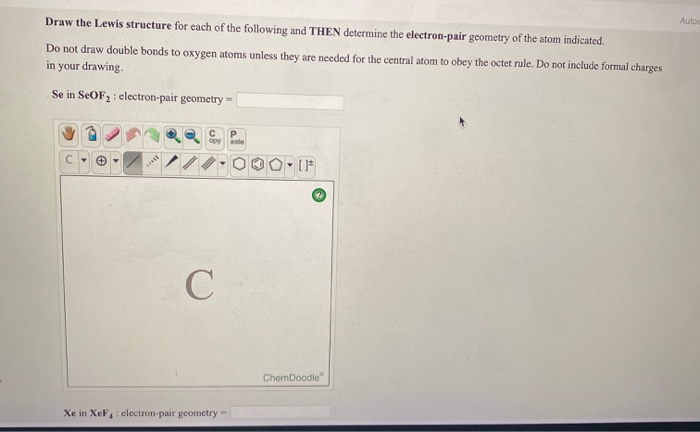
So in part d, what is the mass of one mole of water? Well, it'll be the summed mass of the g of the elements in one mole of water, Or 18.016 g per mole of water. To get the grams of oxygen in one mole of water, 16.00 g oxygen. 
One mole of water will contain one mole of oxygen, which we can then multiplied by the molar mass of oxygen. In order to get the grams of hydrogen in one mole of water, 2.016 g. What are the masses of hydrogen And oxygen in one mole of water? Well, if we have one mole of water, we have two moles of hydrogen we can multiply the two moles of hydrogen by the molar mass of hydrogen. If we have one mole of the compound, then we would have two moles of hydrogen and one mole of oxygen, Part C. These sub scripts can also be used for moles. The two on the hydrogen means that there are two atoms of hydrogen And no subscript on oxygen means that there's one atom of oxygen in one molecule of H 20. This can be determined by the looking at the sub scripts. How many hydrogen and oxygen atoms are present in one molecule of H 20. Here again is one of those ridiculously long questions with three long parts, which again very easily could be separated into three or more questions. Iho throrotical Mc d ol tne roactan Includlng Unil' lntiGio Fumhen otmomiol cruce acelnmirachon Inar Yo4 Kolntcn, ahoxlequnbon: Yn Vnilt Tv Mreuc #humumaleloliou crude ncalan nophe AclunMannenconen uventhouan tou4or ilpobabi as6uplan Ihuly ulwua mJua Wten cukuuluiq Iclds 0 cnena Dccuas AniAGnn Fumhenolmols Iiss nrd wc Jhinro Iro TAM n pun ecobamncarenImal Wscain "qualiont Yith unltt Wenmi Calulald 'ha Parcenl tcd 0l Ino pua acC nmnopher Yleld undehoxalI cjunlony and Icludc oll unl Locnnanneni Vee your tneorellcal (2 pls ) Why Cant you cukcuale Iho yicd ol & reacticn Iukno IFu m3y ohe Koducilo bn min Inilica (eanent $ are in this sample?.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
